Early Detection of Rectal Cancer: Warning Signs You Should Not Ignore
Rectal cancer is a type of cancer that begins in the rectum, and it is one of the most common forms of colorectal cancer. Early-stage rectal cancer often presents with no or subtle symptoms, making it difficult for patients to recognize the disease in its initial stages. When rectal cancer is diagnosed at an advanced stage, treatment becomes more complex, and the chances of successful recovery decrease. Therefore, early detection of rectal cancer is crucial for better treatment outcomes.
Dr. Sandeep Nayak, an ace surgical oncologist in India, states, “Catching rectal cancer early allows us to use less invasive treatments and significantly improve the chances of recovery.”

Early detection of rectal cancer involves recognizing warning signs, understanding risk factors, and seeking professional evaluation if symptoms develop. At MACS Clinic, Dr. Sandeep Nayak offers advanced rectal cancer treatment in Bangalore, combining state-of-the-art technology with personalized care. By focusing on early diagnosis, he ensures that patients receive timely intervention, which can make all the difference in the effectiveness of their treatment, improving their quality of life and long-term health.
Why Early Detection of Rectal Cancer Matters
Early-stage rectal cancer is often treatable with less aggressive interventions. When detected early, the chances of a complete recovery increase dramatically, and patients have access to a wider range of treatment options. In contrast, if rectal cancer is discovered at a later stage, it may have spread to other parts of the body, requiring more intensive treatments such as chemotherapy or radiation therapy.
Dr. Sandeep Nayak explains, “The earlier we catch rectal cancer, the more treatment options we have, and the better the prognosis.” This highlights the importance of screening and being vigilant about any changes in your health, especially if you are at higher risk.
Common Warning Signs You Should Not Ignore
Being aware of the symptoms associated with rectal cancer is key to early detection. While other conditions can also cause some of these signs, it’s essential to seek medical advice if they persist:
- Changes in bowel habits:
Frequent diarrhea or constipation, or a feeling that your bowel doesn’t empty completely.
- Blood in stool:
Bright red blood or dark, tarry stools can indicate bleeding in the digestive tract.
- Unexpected weight loss:
Losing weight without trying can be a sign of various cancers, including rectal cancer.
- Fatigue:
A constant state of tiredness that doesn’t improve even with rest.
- Abdominal pain:
Persistent cramps or bloating in the stomach area.
If you experience any of these symptoms for an extended period, don’t wait; consult an oncologist to rule out potential problems or catch them early.
How Early Detection Improves Treatment Outcomes
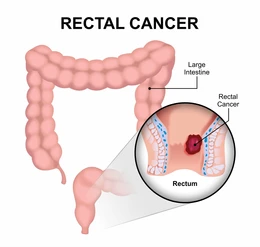
When rectal cancer is detected early, the chances of successful treatment increase significantly. This is because early diagnosis enables:
- Less invasive surgery: Smaller tumors are easier to remove, often with minimally invasive surgery.
- Better chances of survival: Since the disease has not spread too much, the probability of surviving increases.
- Fewer complications: With early treatment, there will be less possibility of developing any complications during and after surgery.
- More options of treatment: It will be possible for the patient to choose whether to undergo surgery, chemotherapy, or radiotherapy, depending on the stage of cancer.
According to Dr. Sandeep Nayak, a distinguished surgical oncologist in Bangalore, India, “By detecting cancer early, we give patients more choices for treatment, which improves both outcomes and quality of life.”
Who Is at Higher Risk of Rectal Cancer?
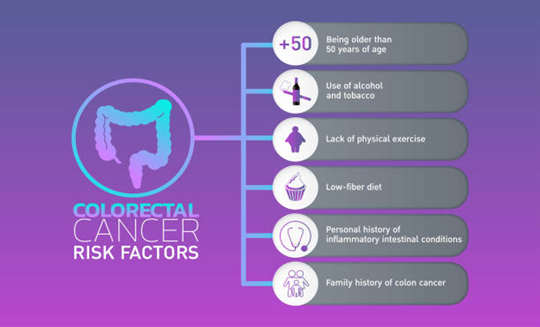
Some factors increase the likelihood of developing rectal cancer, such as:
- Age: Risk increases after the age of 50.
- Family history: A family history of colorectal cancer or polyps increases your risk.
- Diet: High intake of red or processed meat and low fiber foods may increase your chances of getting rectal cancer.
- Personal history: History of having inflammatory bowel diseases, including Crohn’s disease and ulcerative colitis.
- Lifestyle factors: Smoking, excessive alcohol use, and lack of physical activity.
Some risk factors can’t be changed, while others may be controlled by adopting a healthy lifestyle and staying on top of screenings, which can significantly reduce the likelihood of developing rectal cancer.
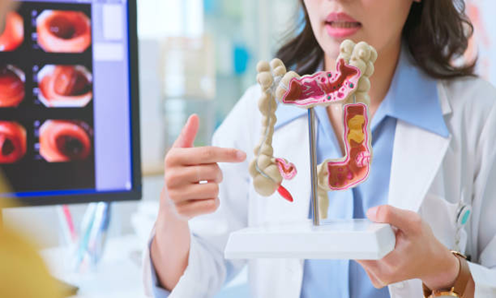
Screening Tests for Early Detection of Rectal Cancer
Screening tests are essential for early detection, especially for individuals at higher risk. Common tests include:
- Colonoscopy:
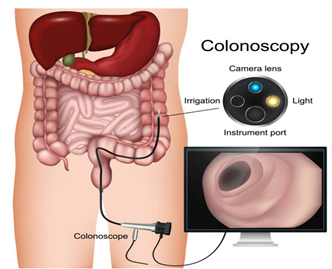
This procedure is highly common and one of the best screening tests used to diagnose rectal cancer and even precancerous polyps.
- Stool Tests:
Stool sample tests, such as FIT or FOBT, help detect blood in the stool, an indication of cancer.
- CT Colonography:
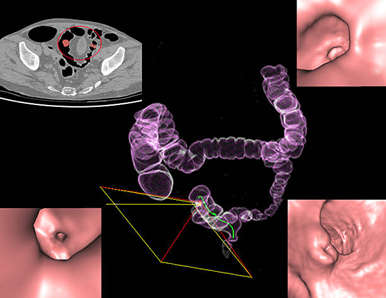
A non-invasive imaging test that creates detailed pictures of the colon and rectum.
- Sigmoidoscopy:
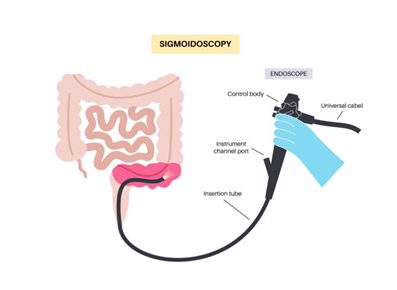
A less extensive form of colonoscopy, sigmoidoscopy, is done to examine the lower part of the colon and rectum.
Screening is highly recommended for individuals over 50 years of age or at high risk. It can detect cancer before symptoms appear, allowing for earlier and more effective treatment.
Tips to Reduce the Risk of Rectal Cancer
Various tips could help one prevent rectal cancer by leading a healthy lifestyle:
- Eat a healthy diet: This includes a diet rich in fruits and vegetables as well as fiber and low in red and processed meat.
- Exercise regularly: Physical activity helps you maintain a healthy weight, hence reducing the risk of cancer.
- Avoid smoking and drinking excessively: These habits will increase the risk of getting rectal cancer.
- Screening: You should screen for early detection through a colonoscopy or a stool test.
- Healthy body weight: Being obese increases the risk of cancer, so maintaining a healthy weight can reduce your risk.
According to Dr. Sandeep Nayak, “Taking simple but important steps to maintain a healthy lifestyle as well as screening often would help prevent rectal cancer.”
Frequently Asked Questions
1. What are the early signs of rectal cancer?
2. Is rectal cancer curable?
3. What screening tests are available for rectal cancer?
4. How often should I be screened for rectal cancer?
5. Is rectal cancer hereditary?
Reference links:
https://www.cancer.org/cancer/types/colon-rectal-cancer/detection-diagnosis-staging.html
Disclaimer: The information shared in this content is for educational purposes and not for promotional use.