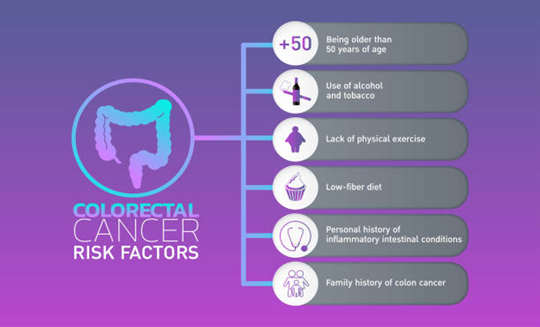
Is a Painless Lump Always Cancer
Painlessness does not make a lump safe to ignore and it does not make it cancerous either. Most painless lumps are benign. Lipomas, sebaceous cysts, fibroadenomas and reactive lymph nodes are all painless and none of them are malignant. What raises concern is not the absence of pain but the feel of the lump, where it sits, how fast it’s growing and whether it’s changing. Those four things together determine whether urgent investigation is needed, not whether it hurts.
According to Prof. Dr. Sandeep Nayak, Surgical Oncologist in India, “pain is a poor indicator of cancer. Some of the most serious lumps are completely painless. What I look at is the feel, the location and whether it’s changing. Those are the real indicators.“
Have a lump you’re uncertain about and want a specialist clinical assessment?
What Makes a Painless Lump More Likely Benign?
Most painless lumps fit recognisable benign patterns that an experienced clinician can identify on examination and ultrasound without needing immediate biopsy.
- Soft and Mobile: Soft, smooth, freely mobile lumps with a clear tissue plane underneath are almost always lipomas and cancer diagnosis is not the clinical priority for a lump that has been stable, non-tender and completely unchanged in size for months.
- Skin Surface Lump: A lump on the skin with a visible central punctum and slow growth over years is a sebaceous cyst, a benign blocked gland that only needs removal if it repeatedly becomes infected or causes discomfort.
- Post-Infection Node: A soft lymph node appearing after a throat infection, dental abscess or skin infection is the immune system doing its job and in most cases it settles completely within two to four weeks without investigation.
- Young Woman, Breast Lump: Firm, smooth, mobile breast lumps in women under 30 are most likely fibroadenomas. Ultrasound confirms this and most are managed with observation rather than surgery unless they grow or cause concern.
Benign lumps share a pattern of softness, clear mobility and stable size over time that fits a known non-malignant diagnosis on examination.
What Features in a Painless Lump Raise Concern for Cancer?
Certain characteristics in any painless lump shift clinical priority from observation to urgent investigation.
- Hard and Doesn’t Move: A lump that feels hard, has irregular edges and resists movement when pressed is clinically concerning regardless of pain and robotic cancer surgery or conventional resection for confirmed malignancy produces far better outcomes when investigation happens promptly rather than after months of delay.
- Growing Over Weeks: Visible size increase over two to four weeks is a more reliable indicator of malignant biology than pain level ever is, and any growing lump warrants ultrasound and biopsy regardless of whether the patient feels any discomfort from it.
- High-Risk Location: Neck, axilla, breast, groin and testis carry higher malignancy probability than lumps elsewhere and any new lump in these sites in an adult over 40 needs imaging and specialist review without delay.
- Accompanying Symptoms: Weight loss, night sweats, persistent fatigue or hoarseness alongside a painless lump points toward a serious systemic or malignant cause and changes the clinical picture significantly regardless of how innocent the lump looks on the surface.
Any lump that is hard, growing, fixed or sitting in a high-risk location needs specialist assessment before a benign diagnosis is accepted and for more on how cancer is confirmed, our blog on cancer diagnosis covers this in detail.
Why Choose Dr. Sandeep Nayak for Lump Treatment and Cancer Surgery?
Dr. Sandeep Nayak brings 24 years of surgical oncology experience, DNB qualifications in Surgical Oncology and General Surgery and a fellowship in Laparoscopic and Robotic Onco-Surgery to lump assessment and cancer surgery across all anatomical sites at KIMS Hospital, Bangalore. He heads Oncology Services across Karnataka with originator credits for RABIT, MIND and L-VEIL techniques and over 25 published clinical studies. Patients with uncertain lumps wanting a clear specialist assessment before any surgical decision are seen here with every case reviewed through tumour board.
Call +91 8104310753 to book your consultation.
Frequently Asked Questions
Are all painless lumps a sign of cancer?
Most painless lumps are benign conditions like lipomas, cysts or reactive lymph nodes. A painless lump becomes concerning when it is hard, fixed, growing or in a high-risk location.
What type of painless lump needs urgent investigation?
A hard, irregular, fixed or growing lump in the neck, breast, axilla, groin or testis in an adult over 40 warrants urgent ultrasound and biopsy regardless of pain level.
Can cancer be completely painless?
Many cancers including lymphoma, thyroid cancer, breast cancer and metastatic neck disease present as completely painless lumps, which is why pain level is not used to judge clinical urgency.
How is a benign lump distinguished from a malignant one?
Clinical examination, ultrasound and where indicated FNAC or core biopsy are used together to distinguish benign from malignant lumps before any treatment decision is made.
References
- National Cancer Institute — Cancer Symptoms and Signs
- World Health Organization — Cancer Early Detection
- Disclaimer: The information shared in this content is for educational purposes and not for promotional use.